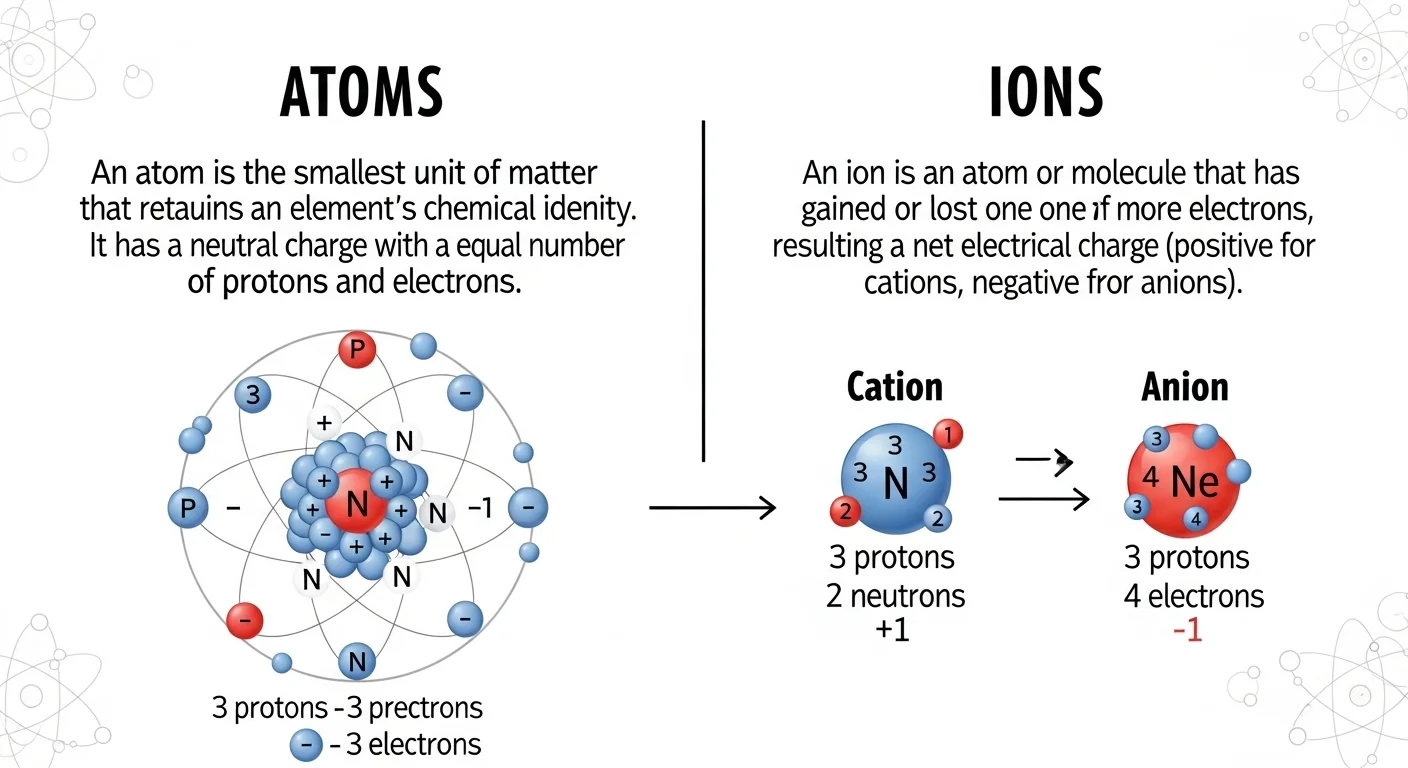
In a small science class, a teacher held a piece of salt and a piece of metal. She said both are made of tiny particles. These tiny parts are called atoms. But then she added something new—ions. The students looked confused. What is the difference between atoms and ions? She explained:
that atoms are neutral, while ions carry charge. This simple idea helps us understand many things in daily life, like why salt dissolves in water or why batteries work. The difference between atoms and ions is very important in science.
In fact, the difference between atoms and ions helps us understand chemistry, electricity, and even life. So, knowing their differences makes learning easier and more fun.
Key Difference Between the Both
Definition/meaning of Atoms:
Atoms are neutral particles.
Definition/meaning of Ions:
Ions are charged particles.
Why Is Their Difference Necessary to Know?
Knowing the difference helps students understand chemical reactions. Experts use this idea in medicine, energy, and industry. It helps in making drugs, building batteries, and cleaning water. So, it is useful for both learning and real life.
Pronunciation
- Atom
- US: /ˈæt̬.əm/
- UK: /ˈæt.əm/
- Ion
- US: /ˈaɪ.ɑːn/
- UK: /ˈaɪ.ɒn/
Now that we know the basics, let us explore the main differences in detail.
10 Differences Between Atoms and Ions
1. Charge
- Atom: Neutral (no charge)
🔴 Example: Hydrogen atom
🔴 Example: Oxygen atom
Sentence: An atom has equal protons and electrons. - Ion: Has charge (positive or negative)
🟣 Example: Na⁺
🟣 Example: Cl⁻
Sentence: An ion forms when electrons are lost or gained.
2. Number of Electrons
- Atom: Equal to protons
🔴 Example: Carbon atom
🔴 Example: Neon atom
Sentence: Electrons match protons in atoms. - Ion: Not equal to protons
🟣 Example: Ca²⁺
🟣 Example: O²⁻
Sentence: Ions have more or fewer electrons.
3. Stability
- Atom: May be unstable
🔴 Example: Sodium atom
🔴 Example: Chlorine atom
Sentence: Some atoms react to become stable. - Ion: More stable
🟣 Example: Na⁺
🟣 Example: Cl⁻
Sentence: Ions often reach stable states.
4. Formation
- Atom: Exists naturally
🔴 Example: Helium atom
🔴 Example: Gold atom
Sentence: Atoms are basic units of matter. - Ion: Formed by gain/loss of electrons
🟣 Example: K⁺
🟣 Example: S²⁻
Sentence: Ions form during reactions.
5. Role in Reactions
- Atom: Starts reactions
🔴 Example: Iron atom
🔴 Example: Zinc atom
Sentence: Atoms take part in reactions. - Ion: Helps reactions continue
🟣 Example: H⁺
🟣 Example: OH⁻
Sentence: Ions help carry charge in reactions.
6. Electrical Conductivity
- Atom: Does not conduct
🔴 Example: Noble gases
🔴 Example: Solid sulfur
Sentence: Atoms alone do not carry charge. - Ion: Conducts electricity
🟣 Example: Na⁺ in water
🟣 Example: Cl⁻ in solution
Sentence: Ions help electricity flow.
7. Size
- Atom: Normal size
🔴 Example: Neutral oxygen
🔴 Example: Neutral magnesium
Sentence: Atoms have fixed size. - Ion: Size changes
🟣 Example: Na⁺ (smaller)
🟣 Example: Cl⁻ (larger)
Sentence: Ions change size after charge.
8. Energy State
- Atom: Higher energy
🔴 Example: Reactive atoms
🔴 Example: Free radicals
Sentence: Atoms may have more energy. - Ion: Lower energy
🟣 Example: Stable ions
🟣 Example: Closed-shell ions
Sentence: Ions are often stable.
9. Presence in Nature
- Atom: Found in elements
🔴 Example: Oxygen gas
🔴 Example: Nitrogen gas
Sentence: Atoms make elements. - Ion: Found in compounds
🟣 Example: Salt (NaCl)
🟣 Example: Calcium chloride
Sentence: Ions form compounds.
10. Example in Daily Life
- Atom: Air particles
🔴 Example: Oxygen
🔴 Example: Nitrogen
Sentence: Atoms are everywhere. - Ion: Salt in water
🟣 Example: Na⁺
🟣 Example: Cl⁻
Sentence: Ions dissolve in water.
Nature and Behaviour
Atoms are neutral and simple. They try to become stable. Ions are charged and active. They move easily in water and help reactions happen fast.
Why People Get Confused About Their Use
People think atoms and ions are the same because both are tiny particles. Also, both come from elements. But the main confusion is about charge. Atoms are neutral, while ions are not.
Table: Difference and Similarity
| Feature | Atom | Ion | Similarity |
| Charge | Neutral | Charged | Both are particles |
| Electrons | Equal to protons | Not equal | Both have electrons |
| Stability | Less stable | More stable | Both seek stability |
| Role | Basic unit | Reactive form | Both in reactions |
Which Is Better in What Situation?
Atoms are better when we talk about basic structure. They help us understand matter. They are used in studying elements and molecules.
Ions are better in reactions and solutions. They help in electricity flow. They are useful in batteries, medicine, and water treatment.
How Are the Keywords Used in Metaphors and Similes?
🟣 “He is like an atom, calm and balanced.”
🟣 “Her mood changed like an ion, full of charge.”
🟣 “A group without change is like neutral atoms.”
🟣 “Energy in the team spread like ions in water.”
Connotative Meaning
- Atom: Neutral (simple, basic)
Example: “He is the atom of the group.” - Ion: Positive or negative (active, charged)
Example: “She entered with ionic energy.”
Idioms or Proverbs
🟣 “Small as an atom” – very tiny
Example: “The error was small as an atom.”
🟣 “Full of charge like an ion” – very active
Example: “He was full of charge like an ion.”
Works in Literature
🟣 The Atom (Science, Isaac Asimov, 1965)
🟣 Ion (Philosophy, Plato, Ancient Greece)
Movies Related to Keywords
🟣 The Atom Man vs Superman (1950, USA)
🟣 Ion Storm (2001, USA)
Frequently Asked Questions
1. What is the main difference between atoms and ions?
Atoms are neutral, ions are charged.
2. How do ions form?
By gaining or losing electrons.
3. Are all atoms stable?
No, some atoms react to become stable.
4. Where are ions found?
In salts, solutions, and living cells.
5. Why are ions important?
They help in electricity and chemical reactions.
How Are Both Useful for Surroundings?
Atoms form everything around us. They build air, water, and solids. Ions help in many processes like nerve signals, water cleaning, and battery power. Both are very important for life and technology.
Final Words
Atoms are the base of matter. Ions are their active form. Together, they explain how the world works.
Conclusion
The difference between atoms and ions is simple but very important. Atoms are neutral and basic, while ions carry charge and are active. This difference helps us understand chemistry and daily life. From salt in food to energy in batteries, ions play a key role. Atoms help us see the structure of matter, while ions show how reactions happen. By learning this concept, students and experts can better understand science and its real-world uses. So, always remember—atoms build the world, and ions keep it moving.

Hi! I am Arshad Ullah presently working as linguist in Punjab Education Department. I have done MA in English Literature while M.Phil in Applied Linguistics. I have taught creative writing to the post graduation classes for 15 years. Presently I am working as content writer, and offering classes for blog writing.